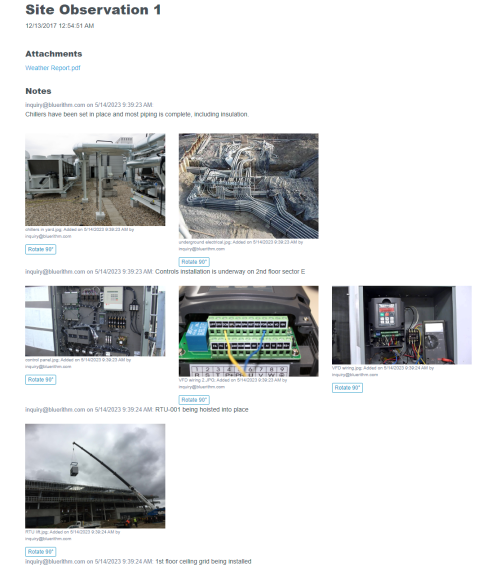
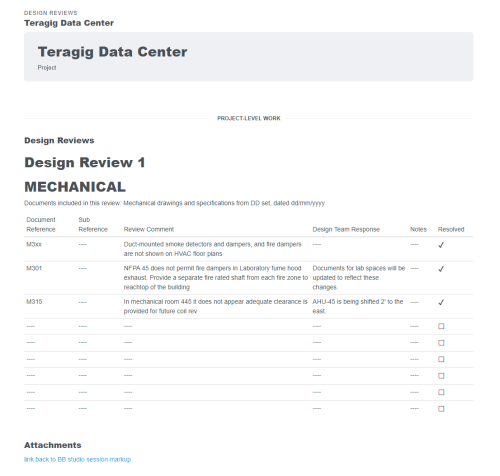
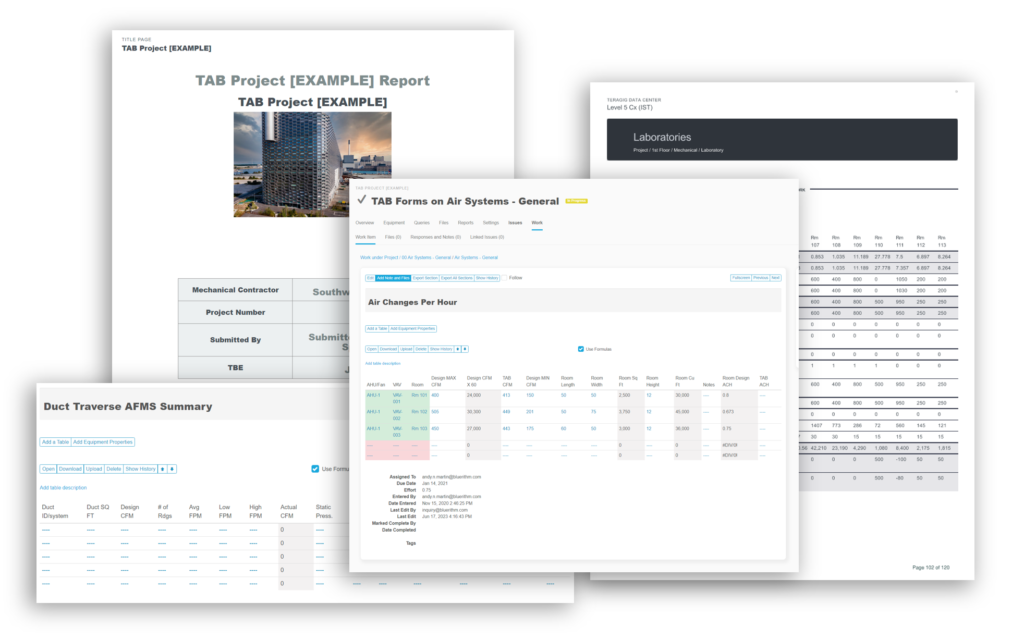
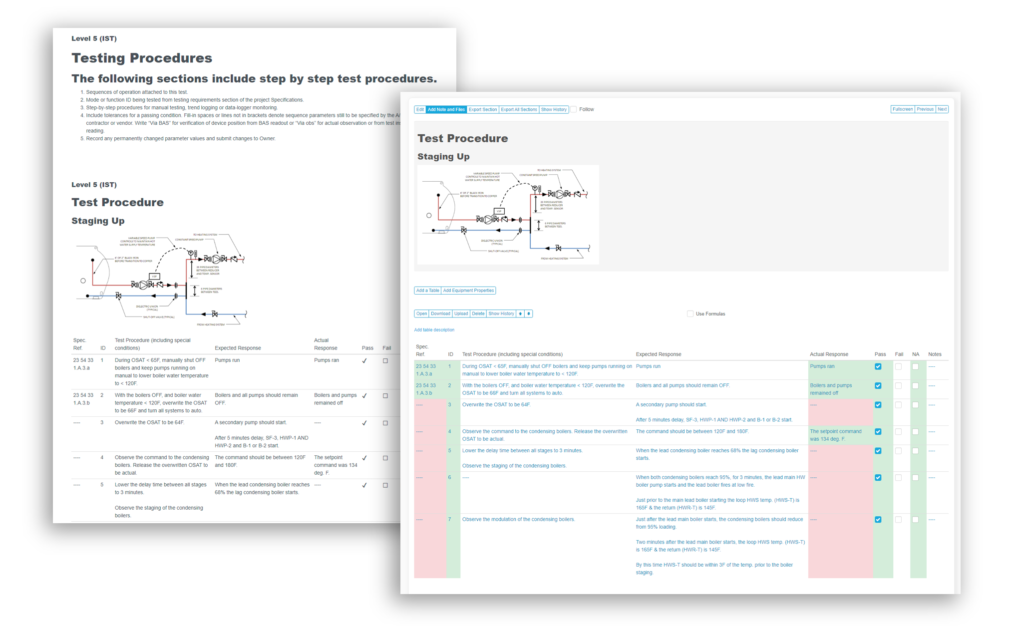
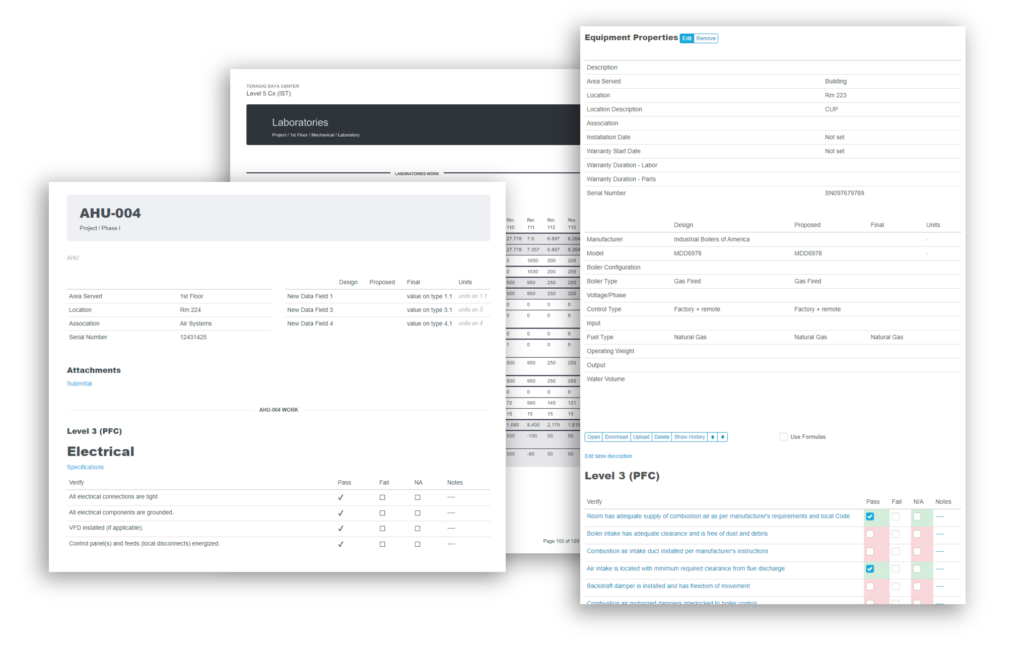
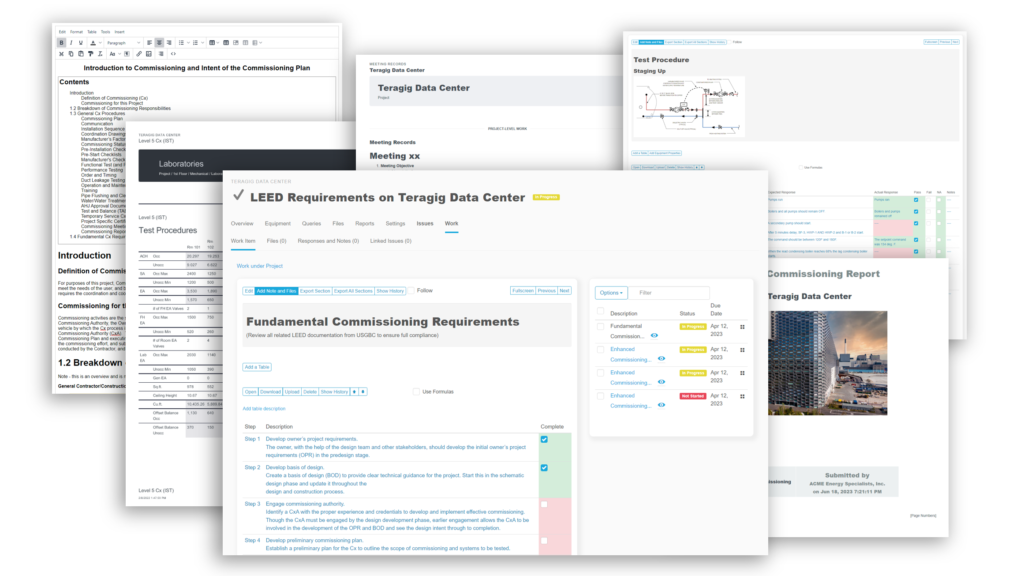
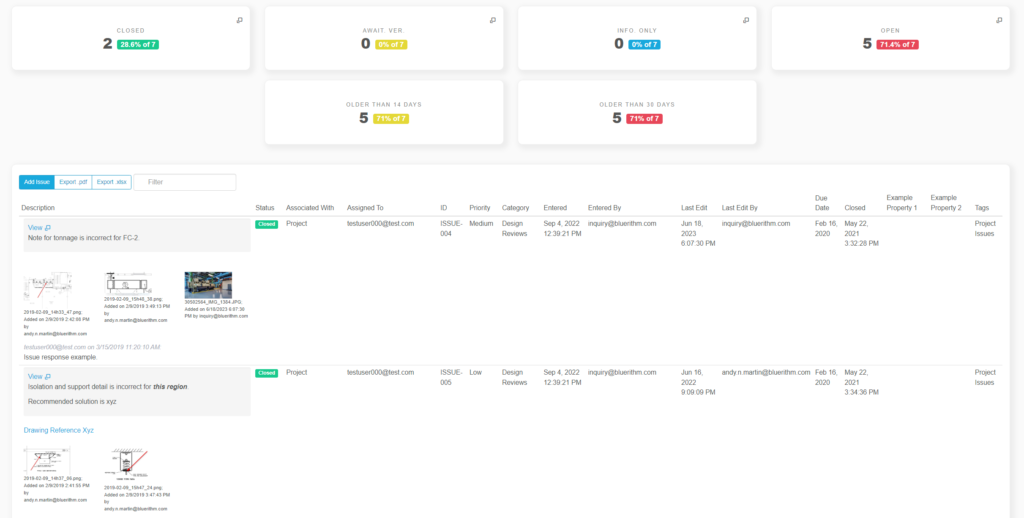
Recently, the JB&B Field™ team decided to move its process to the cloud with Bluerithm. Today, JB&B Field™ uses Bluerithm to streamline progress updates, test scripts, and deficiency tracking—all aspects of their comprehensive workflow—into a single interface. Since launching, the team has reduced redundant project communications, improved transparency, and saved considerable time—all while retaining the stringent process they’re known for.